| Female Sex Enhancement Pharmaceutical Powder CAS 167933-07-5 Flibanserin |
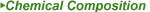 |
| Female Sex Enhancement Pharmaceutical Powder CAS 167933-07-5 Flibanserin |
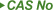 |
| 167933-07-5 |
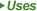 |
Female Sex Enhancement Pharmaceutical Powder CAS 167933-07-5 Flibanserin
Product Name: Flibanserin
Flibanserin CAS No.: 167933-07-5
Flibanserin EINECS No.:
Flibanserin Molecular formula: C20H21F3N4O
Flibanserin Molecular weight: 390.4
Flibanserin Content: 98%
Flibanserin Packing: 1KG/Bag
Flibanserin Appearance: White powder
Flibanserin Grade: Pharmaceutical grade
Usage:a drug being studied as a non-hormonal treatment for pre-menopausal women with hypoactive sexual desire disorder (HSDD)
Catagory: Pharmaceutical raw material female sex enhance
introduction
HSDD is the most commonly reported form of female sexual dysfunction. HSDD is defined as a persistent or recurrent deficiency or absence of sexual fantasies and desire for sexual activity that causes marked distress or interpersonal difficulty, and which is not better accounted for by a medical, substance-related, psychiatric (e.g., depression) or other sexual condition. The cause of HSDD is believed to involve a multitude of social, psychological and biological factors and may be attributed to a complex interplay of these factors.
Flibanserin is a novel, non-hormonal drug that has been studied in clinical trials for the treatment of HSDD in premenopausal and postmenopausal women. The application submitted to the FDA is for premenopausal women. Flibanserin is believed to work on key neurotransmitters, or chemicals, in the brain that affect sexual desire. More specifically, it is thought that flibanserin corrects an imbalance of levels of these neurotransmitters by increasing dopamine and norepinephrine (both responsible for sexual excitement) and decreasing serotonin (responsible for sexual inhibition). In clinical studies, flibanserin was evaluated for its ability to increase the frequency of satisfying sexual events, increase the intensity of sexual desire and decrease the associated distress women feel from its loss.
function:
A new drug designed to boost sexual desire in women is controversial for some and eagerly awaited by others, but it's hit a potentially serious snag. The drug didn't boost women's desire any more than a placebo in two clinical trials. The Food and Drug Administration posted the clinical trial results on its website today in advance of a committee meeting on Friday, when a panel of experts will vote whether or not to recommend approval of the drug called flibanserin. (The FDA usually follows the recommendations of its expert panels.) Although there was a slight increase in the number of sexually satisfying events flibanserin users had each month, the FDA staff who reviewed the results said the so-called response rate isn't "particularly compelling."
Description:
Part of a class of drugs known as nootropic drugs - or 'smart drugs', Centrophenoxine is one of the original anti-ageing, neuro-energizing drugs.
The word nootropic itself is derived from the Greek words 'noos' or 'mind' and 'tropein', which means bend or turn.
Centrophenoxine is an anti-ageing drug, widely used to increase brain energy. It has been found to be extremely effective in the treatment of brain damage due to old age, stroke or toxic chemicals.
Centrophenoxine is used to enhance the body's defense against free radical damage caused by ageing. It is particularly useful in the treatment of senile dementia, where regular use can prevent further mental deterioration.
Often referred to as an 'ageing reversal drug', Centrophenoxine has been clinically proven to improve memory and increase the speed at which new information is stored and used. It is effective in fighting both cerebral ageing and age-related diseases.
Applications:
A new drug designed to boost sexual desire in women is controversial for some and eagerly awaited by others, but it's hit a potentially serious snag. The drug didn't boost women's desire any more than a placebo in two clinical trials. The Food and Drug Administration posted the clinical trial results on its website today in advance of a committee meeting on Friday, when a panel of experts will vote whether or not to recommend approval of the drug called flibanserin. (The FDA usually follows the recommendations of its expert panels.) Although there was a slight increase in the number of sexually satisfying events flibanserin users had each month, the FDA staff who reviewed the results said the so-called response rate isn't "particularly compelling."
It's called the "little pink pill," a tiny tablet that could have a huge impact on treating female sexual dysfunction. If it's approved, it would become the first drug of its kind on the market.
Competitive Advantage:
1 High quality with competitive price:
1) Standard: Enterprise Standard
2) All PurityˇĂ98%
3) We are manufacturer and can provide high quality products with factory price.
2 Fast and safe delivery:
1) Parcel can be sent out in 24 hours after payment. Tracking number available
2) Secure and discreet shipment. Various transportation methods for your choice.
3) Customs pass rate ˇĂ99%
4) We have our own agent/remailer/distributor who can help us ship our products very fast and safe, and we have stock in there for transferring.
3 We have clients throughout the world:
1) Professional service and rich experience make customers feel at ease, adequate stock and fast delivery meet their desire.
2) Market feedback and goods feedback will be appreciated, meeting customers' requirement is our responsibility.
3) High quality, competitive price, fast delivery, first-class service gain the trust and praise from the customers.
Email:sales09 at ycphar dom com
Skype:sales09_109
Telephone:86-027-50756084
Mobile:86-18872220656
|
 |
Female Sex Enhancement Pharmaceutical Powder CAS 167933-07-5 Flibanserin
Product Name: Flibanserin
Flibanserin CAS No.: 167933-07-5
Flibanserin EINECS No.:
Flibanserin Molecular formula: C20H21F3N4O
Flibanserin Molecular weight: 390.4
Flibanserin Content: 98%
Flibanserin Packing: 1KG/Bag
Flibanserin Appearance: White powder
Flibanserin Grade: Pharmaceutical grade
Usage:a drug being studied as a non-hormonal treatment for pre-menopausal women with hypoactive sexual desire disorder (HSDD)
Catagory: Pharmaceutical raw material female sex enhance
introduction
HSDD is the most commonly reported form of female sexual dysfunction. HSDD is defined as a persistent or recurrent deficiency or absence of sexual fantasies and desire for sexual activity that causes marked distress or interpersonal difficulty, and which is not better accounted for by a medical, substance-related, psychiatric (e.g., depression) or other sexual condition. The cause of HSDD is believed to involve a multitude of social, psychological and biological factors and may be attributed to a complex interplay of these factors.
Flibanserin is a novel, non-hormonal drug that has been studied in clinical trials for the treatment of HSDD in premenopausal and postmenopausal women. The application submitted to the FDA is for premenopausal women. Flibanserin is believed to work on key neurotransmitters, or chemicals, in the brain that affect sexual desire. More specifically, it is thought that flibanserin corrects an imbalance of levels of these neurotransmitters by increasing dopamine and norepinephrine (both responsible for sexual excitement) and decreasing serotonin (responsible for sexual inhibition). In clinical studies, flibanserin was evaluated for its ability to increase the frequency of satisfying sexual events, increase the intensity of sexual desire and decrease the associated distress women feel from its loss.
function:
A new drug designed to boost sexual desire in women is controversial for some and eagerly awaited by others, but it's hit a potentially serious snag. The drug didn't boost women's desire any more than a placebo in two clinical trials. The Food and Drug Administration posted the clinical trial results on its website today in advance of a committee meeting on Friday, when a panel of experts will vote whether or not to recommend approval of the drug called flibanserin. (The FDA usually follows the recommendations of its expert panels.) Although there was a slight increase in the number of sexually satisfying events flibanserin users had each month, the FDA staff who reviewed the results said the so-called response rate isn't "particularly compelling."
Description:
Part of a class of drugs known as nootropic drugs - or 'smart drugs', Centrophenoxine is one of the original anti-ageing, neuro-energizing drugs.
The word nootropic itself is derived from the Greek words 'noos' or 'mind' and 'tropein', which means bend or turn.
Centrophenoxine is an anti-ageing drug, widely used to increase brain energy. It has been found to be extremely effective in the treatment of brain damage due to old age, stroke or toxic chemicals.
Centrophenoxine is used to enhance the body's defense against free radical damage caused by ageing. It is particularly useful in the treatment of senile dementia, where regular use can prevent further mental deterioration.
Often referred to as an 'ageing reversal drug', Centrophenoxine has been clinically proven to improve memory and increase the speed at which new information is stored and used. It is effective in fighting both cerebral ageing and age-related diseases.
Applications:
A new drug designed to boost sexual desire in women is controversial for some and eagerly awaited by others, but it's hit a potentially serious snag. The drug didn't boost women's desire any more than a placebo in two clinical trials. The Food and Drug Administration posted the clinical trial results on its website today in advance of a committee meeting on Friday, when a panel of experts will vote whether or not to recommend approval of the drug called flibanserin. (The FDA usually follows the recommendations of its expert panels.) Although there was a slight increase in the number of sexually satisfying events flibanserin users had each month, the FDA staff who reviewed the results said the so-called response rate isn't "particularly compelling."
It's called the "little pink pill," a tiny tablet that could have a huge impact on treating female sexual dysfunction. If it's approved, it would become the first drug of its kind on the market.
Competitive Advantage:
1 High quality with competitive price:
1) Standard: Enterprise Standard
2) All PurityˇĂ98%
3) We are manufacturer and can provide high quality products with factory price.
2 Fast and safe delivery:
1) Parcel can be sent out in 24 hours after payment. Tracking number available
2) Secure and discreet shipment. Various transportation methods for your choice.
3) Customs pass rate ˇĂ99%
4) We have our own agent/remailer/distributor who can help us ship our products very fast and safe, and we have stock in there for transferring.
3 We have clients throughout the world:
1) Professional service and rich experience make customers feel at ease, adequate stock and fast delivery meet their desire.
2) Market feedback and goods feedback will be appreciated, meeting customers' requirement is our responsibility.
3) High quality, competitive price, fast delivery, first-class service gain the trust and praise from the customers.
Email:sales09 at ycphar dom com
Skype:sales09_109
Telephone:86-027-50756084
Mobile:86-18872220656
|
|
|
|
 |
|
|
|
|
|
 |
| There are no existing companies to distribute selected chemical product |
|
 |
|
|










